Building a Strong Data Integrity Framework: Why It Matters and How to Achieve It
In today’s regulated environments, Data Integrity (DI) isn’t just a compliance checkbox—it’s the backbone of trust, quality, and patient safety. Regulatory bodies like the FDA and HPRA demand that data be complete, consistent, and accurate throughout its lifecycle. Achieving this requires a structured approach aligned with computer system validation principles and modern regulatory expectations.

What Is Data Integrity and Why Is It Critical
Data integrity refers to ensuring that data is attributable, legible, contemporaneous, original, and accurate (ALCOA+). In the pharmaceutical and medical device industries, compromised data can result in regulatory non-compliance, product recalls, and even patient harm. A strong data integrity program—supported by effective computer system validation and computer software assurance (CSA) practices—is essential to mitigate these risks.
For organizations in these sectors, data integrity is critical throughout the entire data lifecycle—from creation to archival and retrieval. It guarantees that data and its related information remain complete, consistent, accurate, and reliable, in line with GxP data integrity requirements. Given that pharmaceutical and medical records serve as evidence of results that can directly impact patient safety and consumer health, maintaining the integrity and completeness of data at every stage is paramount.
The Risk-Based Approach
Modern data integrity strategies prioritize risk-based methodologies, which are also central to GAMP 5 software validation and evolving computer software assurance (CSA) guidance. Instead of treating all systems equally, organizations focus on those with the greatest impact on:
- Product quality
- Patient safety
- Regulatory compliance
This approach aligns with global standards and is widely adopted across CSV services in the pharmaceutical industry, helping reduce the likelihood of costly non-compliance.
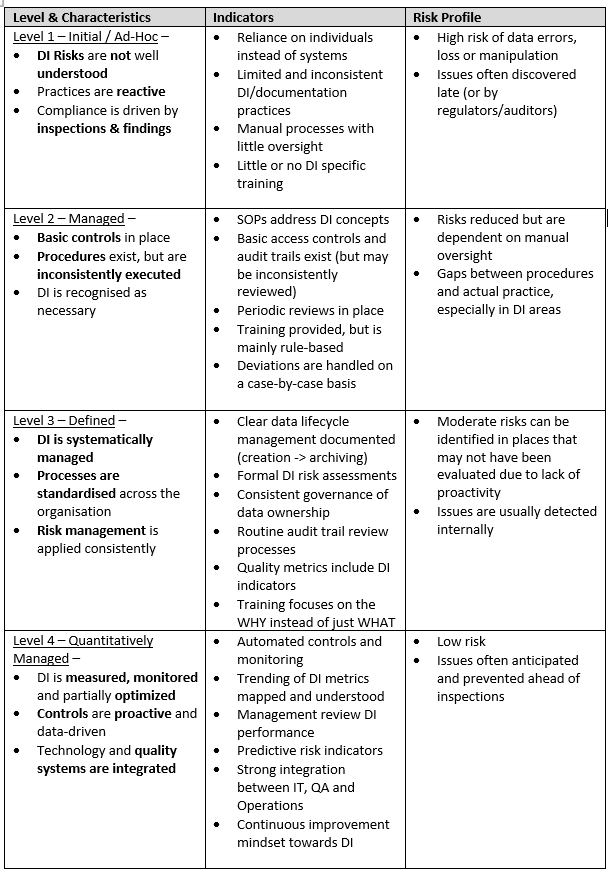
Assessing Maturity: Where Do You Stand?
The International Society for Pharmaceutical Engineering (ISPE) has developed a Data Integrity Maturity Model, which serves as a measurement framework to help organizations assess their level of data integrity implementation. This model complements broader computer system validation strategies by ensuring systems are not only validated but also consistently controlled and monitored.
This model enables companies to determine where they—or individual sites—stand in terms of the complexity and completeness of their data integrity practices.
The maturity model spans from organizations with only a basic understanding of data integrity principles to those that foster a culture of continuous improvement and agility. At the highest level, employees are fully engaged, understand the potential impact of data integrity compliance, can identify possible concerns, and are encouraged to actively contribute to ongoing data integrity initiatives.
By accurately identifying your organization’s position within the maturity model, you can set realistic goals and develop actionable plans to achieve meaningful improvements in data integrity while maintaining alignment with 21 CFR Part 11 compliance expectations.

Key Components of a Data Integrity Program
- Roles & Responsibilities: Clear accountability across QA, IT, manufacturing, and lab teams.
- System Inventory List (SIL): A centralized record of all GxP-relevant systems—critical for transparency, audit readiness, and computer system validation activities.
- GxP Assessment: Evaluating compliance with Good Practices (GMP, GLP, GCP) and GxP data integrity requirements.
- Risk Assessment & Scoring: Identifying high-risk systems and prioritizing remediation in line with GAMP 5 software validation methodologies.
- Data Flow Mapping: Visualizing how data moves across systems to spot vulnerabilities and ensure 21 CFR Part 11 compliance.
- Summary Report: Outlines the goals and remediation actions moving forward, evaluating current DI maturity and validation status.
With this in mind, Dataworks has developed a Data Integrity (DI) Playbook designed to help organizations evaluate their maturity level as part of a broader computer system validation and compliance strategy.
Common Pitfalls
Regulatory audits often uncover:
- Undefined system ownership
- Inadequate audit trail reviews (a key gap in 21 CFR Part 11 compliance)
- Poor access control management
- Missing data governance structures
Avoid these by embedding governance, validation, and continuous improvement into your culture, supported by modern computer software assurance (CSA) approaches.
The Bottom Line
Data integrity is not a one-time project—it’s an ongoing commitment. By adopting a structured, risk-based approach aligned with computer system validation, leveraging frameworks like GAMP 5 software validation, and meeting GxP data integrity requirements, organizations can build resilience, maintain compliance, and protect patient safety.
Ready to strengthen your data integrity and validation approach?
Whether you’re starting your journey or evolving towards CSA and GAMP 5, our experts can help you build a compliant, scalable computer system validation framework tailored to your organisation.
Talk to our validation specialists today
Phone: 051 878 555
Email: team@dataworks.ie
Website: www.dataworks.ie



